Signal processing in the ganglion cell microcircuit
Robert G. Smith
|
The ganglion cell's circuit includes photoreceptors and bipolar
cells which are "vertical components" that transmit signals to
downstream neurons. It also includes "horizontal components",
the horizontal cells and amacrine cells that transmit signals
laterally. One question that we are trying to answer is what is
the function of each cell type, i.e. why does that cell type
exist and what is its function in the circuit. Since the ganglion
cell is the output cell for the retina, we postulate that
each cell type exists to improve the ganglion cell signal in some
specific way. Our hypothesis is that noise generated by the
retinal circuit itself reduces the quality of the visual signal
passed to the brain.
|
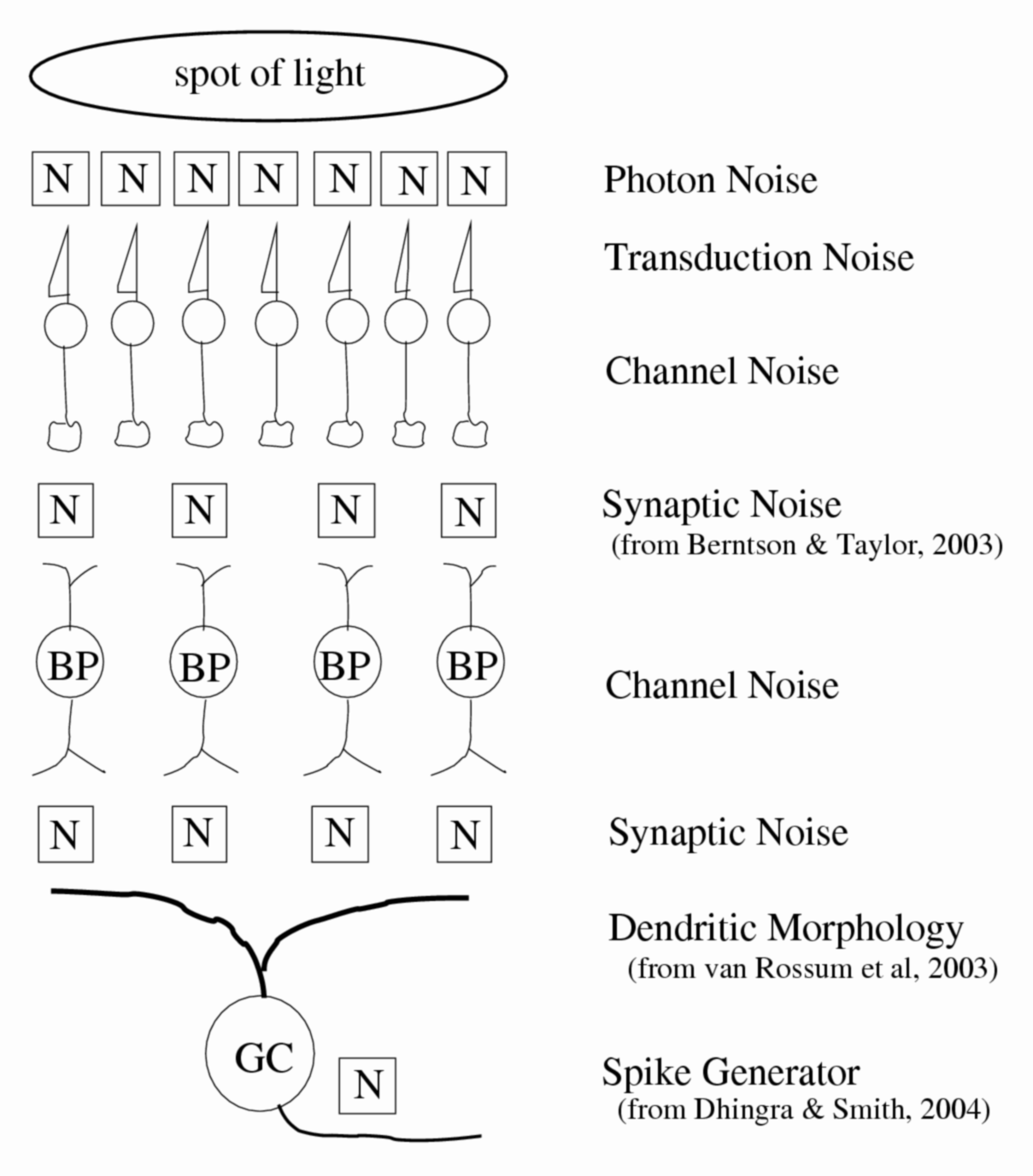
Figure 1. Retinal noise sources. One problem facing the retina is that the visual signals it collects are mixed with noise from various sources. The light collected by photoreceptors contains photon (Poisson) noise. Synapses that transmit the signal from one neuron to the next generate noise from the random fluctuation of vesicles that contain the neurotransmitter released into the synaptic cleft. Voltage-gated and ligand-gated ion channels flicker open and closed randomly.
|
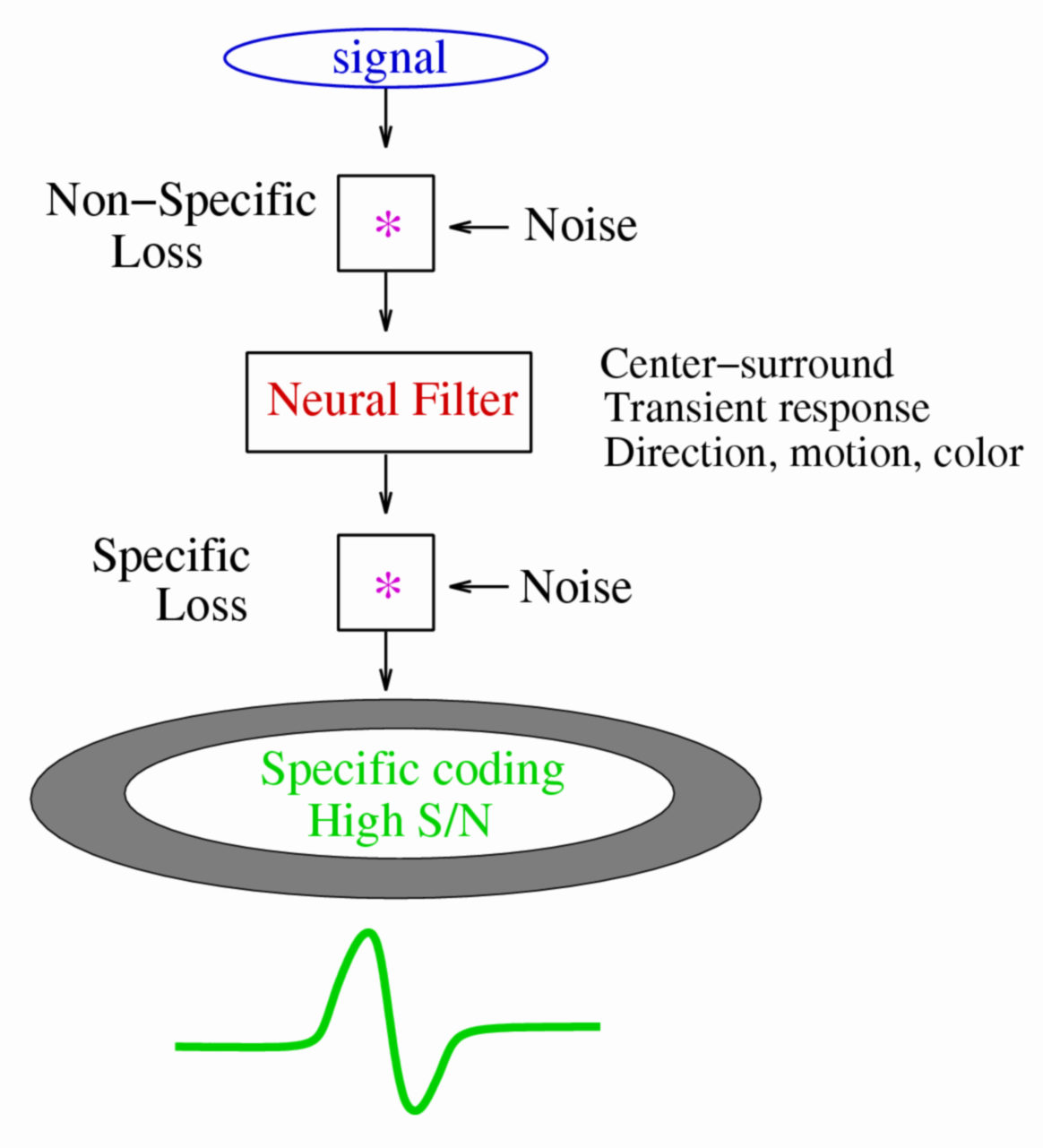
Figure 2. Noise in neural circuit loses information. Top, because noise contains all signal components, when mixed with the signal it causes a "non-specific" loss of information. Middle, a neuron filters the signal it receives, attenuating some signal components. The center-surround antagonism of the receptive field removes the low spatial frequencies, a transient response removes low temporal frequencies, and motion selective response removes static signals. If such a neural filter is linear, it does not result in any information loss because the reverse filter would restore the signal. Bottom, noise mixed with the filtered neural signal causes a "specific" information loss because the components that were attenuated are specifically affected by the noise.
|
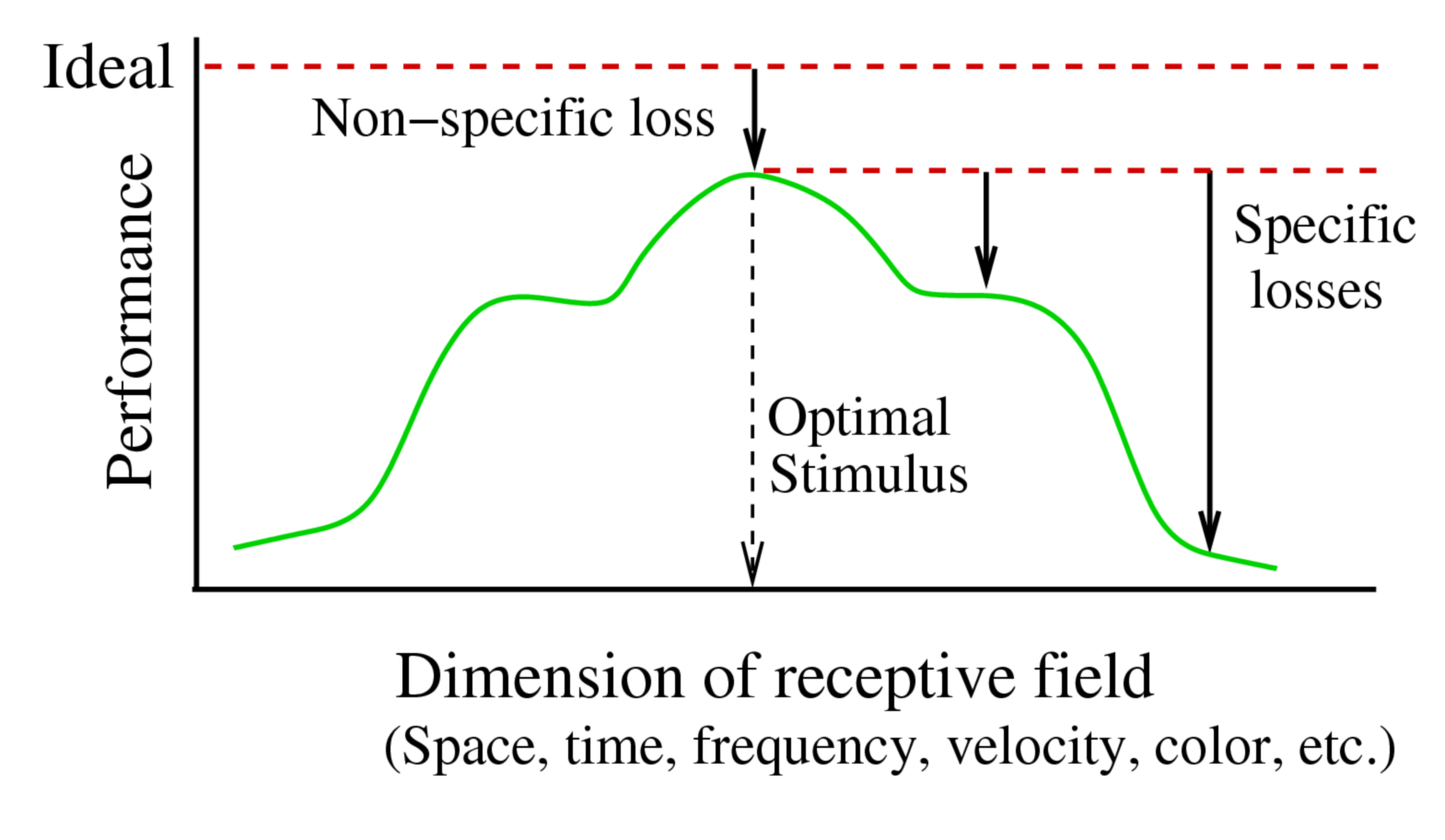
Figure 3. Comparison of Non-Specific and Specific Losses. The non-specific loss originates in uncorrelated noise and so reduces the performance at every point in the receptive field by roughly the same amount. The specific loss originates in the receptive field filter and so differs for every point. To determine the non-specific loss of information, one compares a neuron's performance for the optimal stimulus to the ideal performance of the cone array. To determine the specific loss of information, one compares a neuron's performance for sub-optimal stimuli with the non-specific loss.
|
|
We are currently studying how the retinal ganglion cell circuit processes signals. We can determine the quality of the signal by measuring its signal/noise ratio (S/N). The noise sets the smallest unit of amplitude that the signal can represent. The greater the ratio of signal to noise, the greater the number of gray levels it can represent, and the greater the information it can contain. If the S/N in the path for the visual signal decreases, then some information is lost.
|
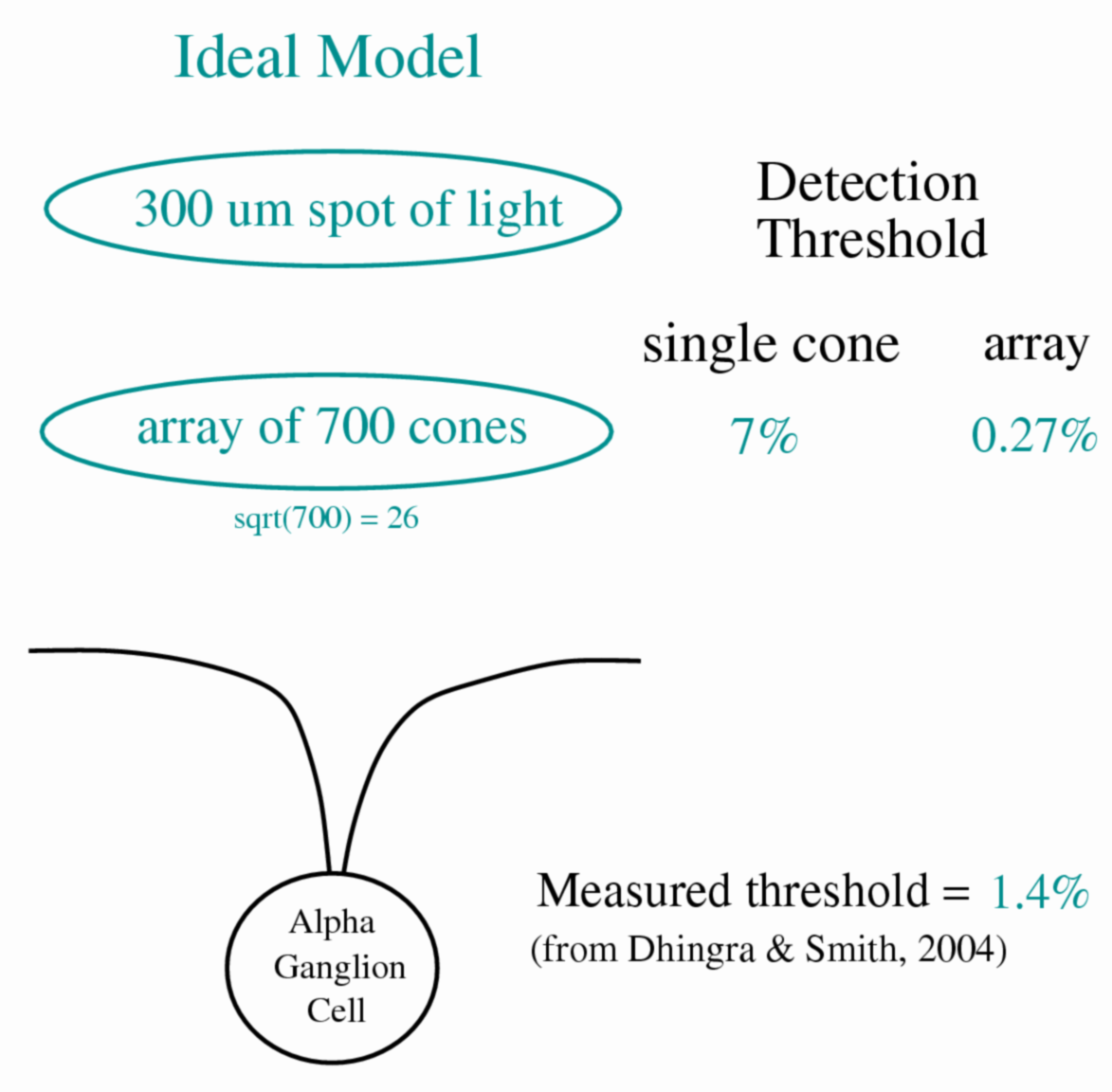 Figure 4. Ideal model of ganglion cell containing only absorption
of light by cones. To determine the "ideal" performance at the
cone outer segment we evaluated the number of photon
isomerizations in the ~700 cones illuminated by a 300 um spot,
calculating the contrast threshold as the contrast required to
generate a response equal to the standard deviation of photon
noise (x1.5).
Figure 4. Ideal model of ganglion cell containing only absorption
of light by cones. To determine the "ideal" performance at the
cone outer segment we evaluated the number of photon
isomerizations in the ~700 cones illuminated by a 300 um spot,
calculating the contrast threshold as the contrast required to
generate a response equal to the standard deviation of photon
noise (x1.5).
A spot of light 300 um in diameter stimulates the centermost portion of the ganglion cell receptive field center, which contains an array of 700 cones. At a background of ~23,000 photons/um2/sec, each cone absorbs photons to generate ~420 rhodopsin isomerizations from a 50 ms flash. Ideal performance, calculated from the standard deviation of the number of photons absorbed, was 0.27%. Real performance of a ganglion cell with the same stimulus was 1.4%, or 5-fold worse.
|
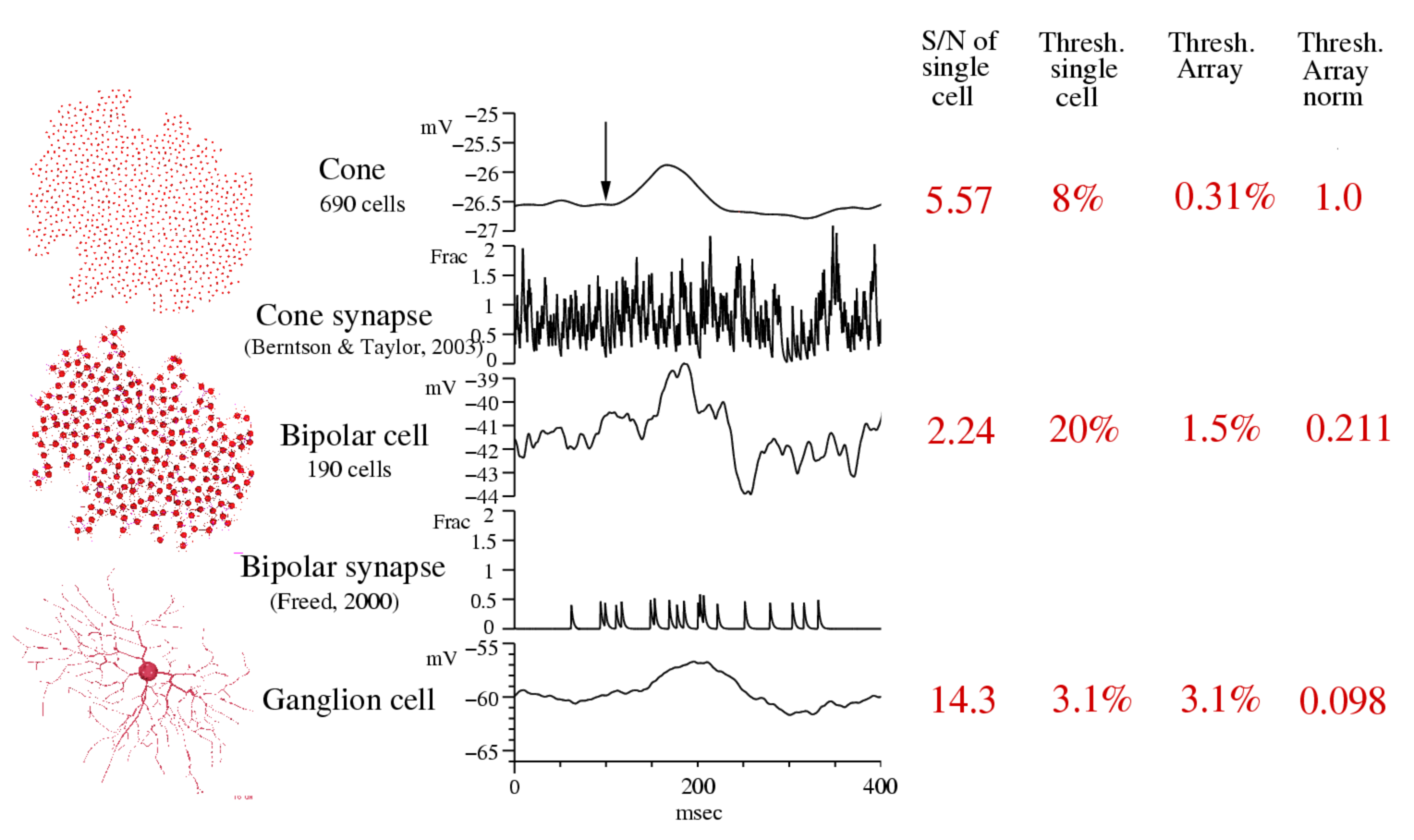
Figure 5. Signal quality in a "practical model" of ganglion cell circuit. To discover the source of the ganglion cell's non-specific information loss, we constructed a compartmental model of a brisk-transient ganglion cell and a simple approximation of its presynaptic circuit that included arrays of cones (n=690) and cone bipolar cells (n=190). Cones included a realistic light response with photon noise and noisy synaptic output. Each cone bipolar collected signals from 4-6 cones through a synapse that included temporal filtering and noise properties. The ganglion cell collected synaptic inputs from all cone bipolars within a threshold distance (10 um) of its dendrites. Synapses had exponential gain (e-fold per 3-6 mV) modulating Poisson release of vesicles (50-100/s). The postsynaptic properties included nonlinear ligand binding modulating a conductance without channel gating noise. The simulation was performed with the Neuron-C simulation language. The S/N ratio of a single cone's response to a dark flashed spot covering the ganglion cell's receptive field was 5.5. The S/N of a single cone bipolar's response was 2.24, and the S/N of the ganglion cell's response was 14. The contrast threshold for the cone (the contrast that exceeded the standard deviation of the noise by a factor of 1.5) was 8%, for the cone bipolar it was 20%, and for the ganglion cell it was 3.1%. The contrast threshold for the array of cones was 0.31%, for bipolar cells, 1.5%. Normalizing the cone threshold to be 1, the cone bipolar array threshold was 5-fold worse, and the ganglion cell threshold was 10-fold worse. With this analysis, we hope to dissect the loss of information in the ganglion cell's circuit, gaining an understanding of the function of each component in the processing of information.
|